Study materials & logistics solutions – reliable, secure, worldwide
Clinical Trial Supply Management by TRIGA-S
We deliver your study materials and samples reliably – handling kit assembly, temperature-controlled sample shipping, and customs clearance. With end-to-end logistics for your IVD studies covered, you can focus on what matters most: your study results.
Because every study is unique, you can book all our services individually, combined, or as a complete package – tailored exactly to your needs.
Your Benefits with TRIGA-S
Reliable Supply
Your trial sites receive study materials on schedule via temperature-controlled transport with full documentation.
Flexible without Minimum Quantities
Whether you need one kit or a thousand, whether planned or at short notice – we adapt to your requirements.
In Compliance with Regulations and Standards
We understand the regulatory requirements for hazardous materials classification, transport regulations, and customs documentation, and apply them correctly to ensure your shipments clear customs smoothly without delays.
Error-Free at Trial Sites
Validated kits and sample panels with verified barcodes and quantities – every shipment runs through a quality inspection before dispatch, reducing errors and returns to a minimum.
One Point of Contact, Clear Responsibilities
Fewer handoffs, faster execution – with us, planning, storage, shipping, and returns management come from a single source.
Our services in detail
Clinical Trial Supply Management
All services at a glance – jump right to your desired topic.
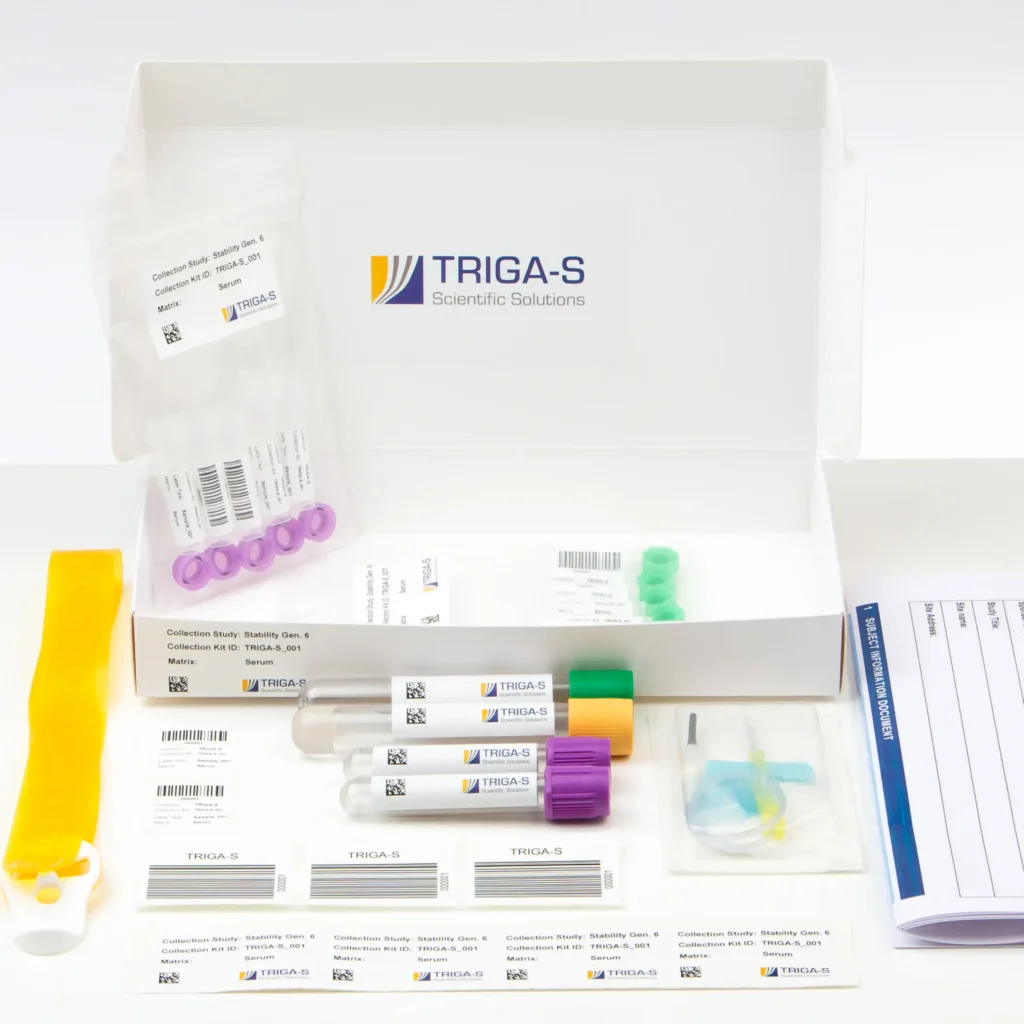
Collection Kit Supply
Every minute counts at trial sites. That’s why we handle kit assembly for you: each collection kit is precisely tailored to your study protocol – with all required components, verified barcodes, and pre-validation. Your trial sites can begin sample collection immediately, without queries or reorders.
- Study-specific kits instead of standard solutions: Whether tubes, needles, or transport containers – we assemble customized kits tailored to your study and application. This makes all the difference, especially in time-sensitive environments such as the emergency room.
- Pre-validation for immediate use: Verified barcodes, controlled quantities, guaranteed completeness and readability – your trial sites can start right away.
Reference Samples & Control Materials
Without standardized reference materials, test results are not comparable. We produce study-specific reference and control materials that enable consistent results across all trial sites.
What we produce for you:
- Study-specific materials for quality assurance and comparability
- Control materials for method validation and calibration
- Sample panels with defined characteristics for benchmarking
Microbiology & Cell Culture
Studies with cell-based assays require specialized laboratory capacity. In our BSL-2 laboratory, we prepare the biological materials your study requires.
Our microbiological services:
- Cultivation and propagation of cell lines and bacterial strains
- Cell counting for defined cell quantities
- Production of cell culture-based reference samples for realistic and reproducible test conditions
- Special requirements for cell-based assays

Logistics Solutions: Worldwide, Secure, Traceable
We manage worldwide shipping of your study materials end-to-end – ensuring temperature requirements are maintained, customs regulations are met, and every shipment is fully documented – for secure sample logistics, on-time delivery, and seamless study operations.
Your study logistics in good hands:
Worldwide Material and Sample Transport
We ensure full chain-of-custody traceability and maintain the integrity and delivery reliability across all transport routes.
Expedited Pickup and Shipping
We meet your timelines, even with unforeseen changes.
Hazardous Materials Logistics
Our packaging and labeling in accordance with applicable regulations ensure the compliance and safety of your shipments.
Customs Clearance, Import Permits, and Study Adjustments
We help you navigate schedule adjustments, changes in sample volume, or shifts in study duration – minimizing delays and keeping costs under control.
Temperature-Controlled Storage of Your Samples and Materials
Study materials and samples often need to be stored for months or years. Throughout this period, quality should be fully maintained. We store your materials and samples under controlled conditions with continuous temperature monitoring and cold chain documentation.
Stable and available:
- Temperature-monitored storage
- Frozen storage (−90 to −60°C, −25 to −15°C), refrigeration (+2 to +8°C), room temperature (+15 to +25°C)
- Short- and long-term storage according to requirements and study duration
- Secured by redundant systems

Ask us About our Full-Service Solutions!
"Global studies demand logistics you can rely on. We cover kit production, temperature-controlled storage, and worldwide sample shipping – always compliant with hazardous materials regulations. Upon request, we run your logistics end-to-end, reducing coordination effort and minimizing errors at handoff points – while saving valuable time."
– Tobias Fröhlich, Head of Logistics
Get in touch
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Email Address
info@triga-s.de
Phone Number
+49 (0) 88 47 – 695 78 0
Address
Mühltal 5, 82392 Habach
Contact Form
You might also be interested in

Study Management and Monitoring
Abbreviations
IVD: In-vitro diagnostics
BSL-2 Laboratory: Contract Laboratory with biosafety level 2


