Traceable. Stable. Ready to use.
Sample Management: Integrity, Traceability, Processing & Panels
Traceable samples, controlled storage, aliquoting, and tailored comparison and control panels: We support your studies exactly where you need us and deliver end-to-end sample management with seamless documentation.
All our services can be booked individually or as a complete package – precisely according to your needs.
Your Samples in Safe Hands
Every sample tells a story. We ensure the quality and integrity of your samples and that their story is seamlessly documented, fully traceable, and audit-ready throughout the entire lifecycle – from receipt, storage, and processing to analysis, retention, and compliant disposal.
Every step in the lifecycle of your samples is meticulously documented, ensuring individual samples are traceable at all times and your processes remain audit-ready.
Depending on requirements, samples are stored at -90 to -60 °C, -25 to -15 °C, +2 to +8 °C, or at room temperature (+15 to +25 °C) – backed by redundant systems to ensure continuous stability and on-time availability.
Our device-compatible labels and barcodes are tailored to your platforms – avoiding reading errors so that laboratory processes function efficiently and reliably.
We aliquot even small volumes and can adapt timings and processes at short notice if needed – valuable sample material is optimally utilized, and you benefit from maximum flexibility.
From collection to storage, processing, and shipping, all steps of study logistics are interconnected with us – ensuring smooth coordination of processes and minimizing handover risks.
Our services in detail
Sample Management: Integrity, Traceability, Processing & Panels
All services at a glance – jump straight to the topic you need.
Sample Administration and Quality Assurance
We handle your entire sample administration, ensuring traceability, controlled storage, and quality assurance. From the first contact with the sample to the final analysis, we keep track of everything – and you stay in control.
Receipt & Documentation
Every sample receives a unique identity as soon as it arrives with us, and we document every step of its lifecycle.
Traceability at every step:
- Seamless documentation: From arrival to your defined purpose, you always know where your sample has been, how it got there, and how it was stored.
- Transparency via LIMS: Our Laboratory Information Management System makes inventory, movements, and status transparent. You can see at a glance what was last recorded.
- From individual samples to large studies: Whether it’s a single sample or a study with several thousand samples – our systems adapt, allowing you to scale flexibly.
Discrepancy Management
Before a sample is entered into our system, we carry out a thorough incoming check to confirm everything is in order.
Quality check on receipt of goods:
- Inspection on arrival: We check every delivery for completeness, sample integrity, conformity, and pre‑analytical requirements.
- Rapid response: If there’s any discrepancy between the delivery and the order, we act quickly and reliably. We document the issue end‑to‑end, inform you immediately, and initiate corrective action – so your study stays on track.
Sample Storage Under Controlled Conditions
Stability starts with the right environment. Some samples need cool conditions; others must be stored deep-frozen. We create the conditions your samples require – and keep them consistent.
Optimal storage conditions:
- Freezer storage (-90 to -60 °C, -25 to -15 °C), refrigeration (+2 to +8 °C), room temperature (+15 to +25 °C): each sample is stored in the environment it needs.
- Short- and long-term storage: From a few days to several years, depending on study duration and client requirements.
- BSL-2: Biological materials such as blood, serum, plasma, or tissue are stored in line with regulatory requirements.
- Redundant Systems: Equipment failure won’t put your samples at risk. Backup systems protect them around the clock.
The Journey of Your Samples at TRIGA-S
Sample Processing and Labelling
From aliquoting to barcoding, we precisely prepare your samples for analysis and ensure that each one remains uniquely identifiable.
Processing
Not every sample arrives ready for analysis. We prepare each sample individually for you, so your analytical testing has optimal conditions.
What we do for you:
- Aliquoting: We divide samples into smaller aliquots so you can run parallel analyses or retain archive material. Each aliquot remains uniquely assigned to the original sample.
- Individual preparation: Diluting, mixing, enriching with defined analytes (e.g., with proteins or metabolites); we assemble your samples according to protocol to represent study-specific matrices and concentration levels.
Labelling and Barcodes
A barcode is the link between a physical sample and a digital data record and must function under all conditions.
Our labeling solutions:
- Precise, device-compatible barcodes: We create labels – including QR codes – for maximum readability, thereby preventing misassignments from the outset.
- Labels for extreme conditions: Our labels withstand moisture, freezing temperatures, and mechanical stress. Designed to remain readable – even after months at -80 °C.
- Preparation of carrier tubes: We label tubes so that automated systems can reliably grasp and scan them.
- Relabelling: To maintain uniqueness and prevent sample loss, we can relabel samples – this also applies to frozen material.
- Troubleshooting for faulty barcodes: Have you received samples with illegible or incorrect codes—or labels damaged by storage conditions (e.g. freezer storage)? We identify the affected samples, correct the labelling, and document the process end-to-end to protect data integrity and throughput.
Sample Panels and Control Materials
Preparation of Sample Panels
A study is only as good as the samples used. Too little variance and you overlook edge cases. Too much variability and your results become difficult to compare.
We assemble panels that precisely match your project requirements:
- Customized compositions: for validations or comparative studies that fit your study design and ensure meaningful and comparable results.
- Homogeneity and quality: to minimize fluctuations and increase the significance of your results.
Provision of Comparison and Control Samples
Reproducible measurements depend on reliable reference materials. On request, we prepare study-specific comparison and control materials for:
- Calibration
- Quality assurance
- Benchmarking
Microbiology and Cell Culture
Some assays require living cells, others defined cell counts or specific culture conditions. We culture, expand, and count cells according to your specifications and produce cell culture-based comparison samples that create realistic test conditions and make your validation meaningful.
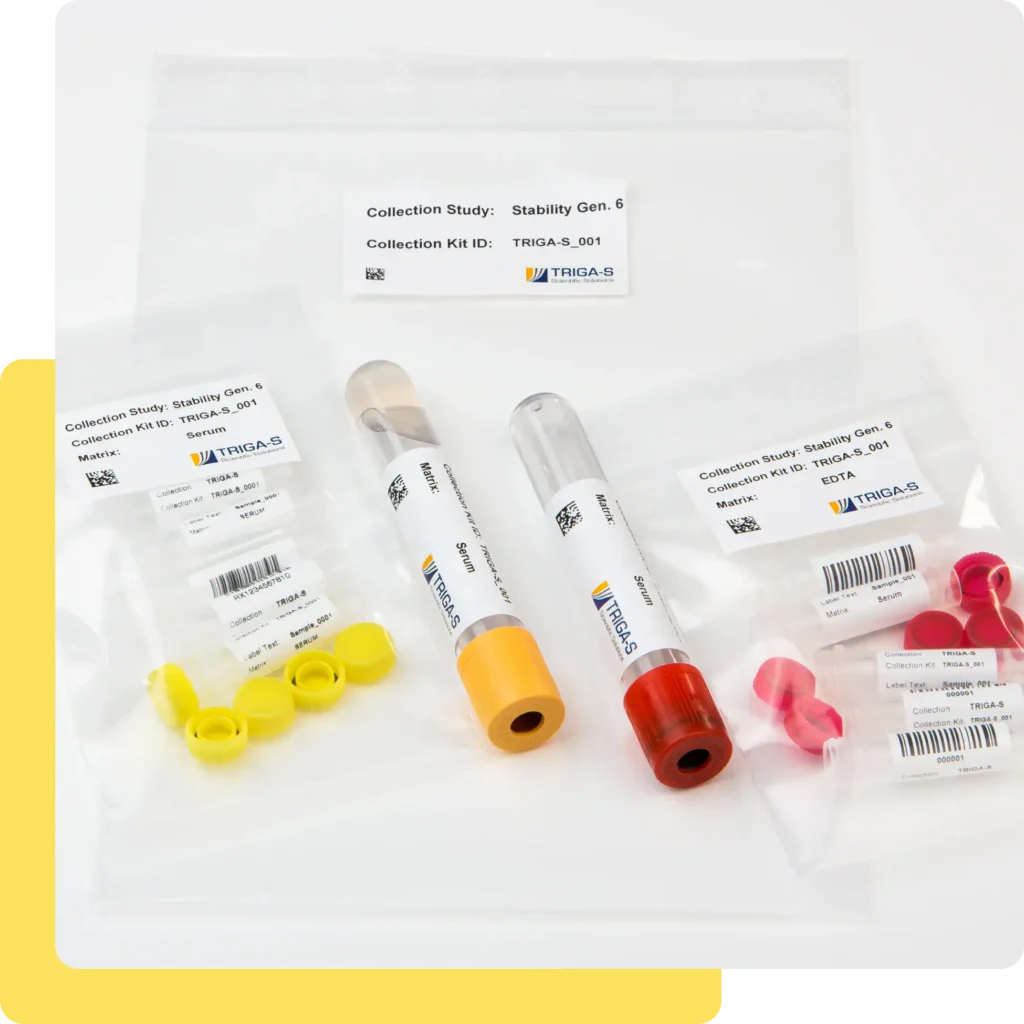
Kit Assembly
Every study succeeds or fails based on sample quality – and this is critically influenced by sample collection. We assemble fully customised collection kits and check every detail before the kits are sent to the study sites:
- Conception and assembly: We put together individual collection kits that are precisely tailored to your study requirements, ensuring a smooth workflow at the study sites.
- Validation before shipping: Barcodes, quantities, components – kit batches are checked to prevent incorrect packing and returns.
- Pre-configured barcodes and tube racks: We prepare barcodes and racks so that your analytical devices can read them reliably. Maximum readability, minimal manual rework.
Sample Collection Studies
Do you need specific samples at a certain disease stage or with defined characteristics? Sometimes it’s not enough to use existing sample material. In these cases, we organize a sample collection study for you – completely, from start to finish.
Planning & Study Design
We define together: Which samples do you need? Quantity, quality, diversity, and pre-analytical criteria (sampling, transport, storage, processing).
Site Selection
We identify the right study sites and coordinate the collaboration.
Regulatory & Documentation
Ethics applications, contracts, patient information: we take care of all documents and ensure legally compliant and smooth processes.
Execution & Quality Control
We accompany sample collection, check quality upon arrival, ensure compliance with pre-analytical requirements, and make sure that you ultimately have the samples you need to cleanly cover your study design and endpoints.

Sample Shipping and Logistics
We deliver your samples safely to their destination with seamless traceability, correct documents, and compliant packaging – adhering to defined temperature and time windows, as well as applicable dangerous goods regulations.
Our logistics services for you:
- Worldwide transport with seamless traceability, so you always know where your samples are. You receive documentation on origin, condition, and transfers for your records and audits.
- We also carry out collection and sample shipping at short notice. Because schedules can be tight, and your study should not be delayed.
- We manage logistics according to dangerous goods regulations with special packaging and labeling. Our trained staff ensure compliance and minimize risks – nationally and internationally.
Integrated Solutions – All From One Partner
International studies require many components. However, you don’t have to coordinate multiple service providers.
We provide complete solutions for your entire sample management: from the assembly of collection kits, to worldwide shipping, to sample administration. One point of contact, one responsibility, maximum efficiency.
"Your samples deserve the best care. Let us work together to develop the optimal solution for your sample integrity. Whether individual samples or international studies – we are your partner."
Get in touch with us
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Email Address
info@triga-s.de
Phone Number
+49 (0) 88 47 – 695 78 0
Address
Mühltal 5, 82392 Habach
Contact Form
You might also be interested in

Sample Management

Study Management and Monitoring
Abbreviations
IVD: In-vitro diagnostics
LIMS: Laboratory Information Management System
BSL-2 Laboratory: Contract laboratory with biosafety level 2


